Admission CTAs
Mason Scientists and Rutgers Cancer Institute of New Jersey collaboration receives U.S. Army’s Breakthrough Award to better inform HER2 breast cancer treatment
A George Mason University and Rutgers Cancer Institute of New Jersey collaboration received the U.S. Army’s Breast Cancer Research Program (BCRP) Breakthrough Award to quickly confirm if an identified HER2 biomarker can indicate success likelihood of personalized breast cancer treatments.
George Mason University’s College of Science announced an exciting $1.33 million collaboration with Rutgers Cancer Institute of New Jersey and Mason’s Center for Applied Proteomics and Molecular Medicine (CAPMM) which taps cancer research diagnostics and treatment strengths within the medical facilities and CAP/CLIA laboratory respectively of the two regional powerhouses.
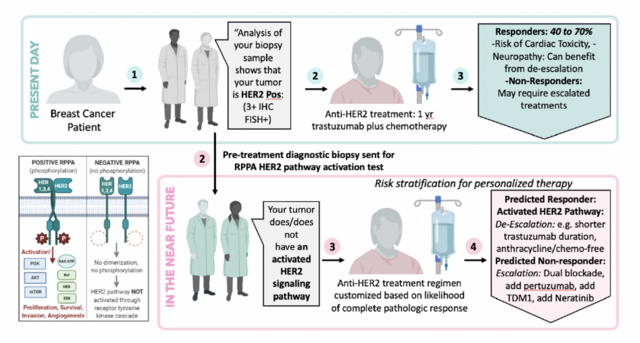
The effort will use a new way to measure biomarkers of the HER2 protein, which is expressed on breast cancer cells. Women who have this protein get specific therapies, now part of the standard of care in both early stage and metastatic settings of breast cancer.
The project’s overall goal is clinical validation, in two independent patient cohorts from the Rutgers Cancer Institute, of a new way for measuring HER2 based on its activation (phosphorylation) and predicting, using only the patient’s diagnostic biopsy, whether or not that patient will respond favorably to the treatment.
The Mason-developed test uses a new Fluidigm laser capture microdissection (LCM), which enables researchers to capture cellular regions of interest from frozen and FFPE tissue sections for downstream analysis using DNA and RNA sequencing, real-time PCR or mass spectrometry analysis. The test is designed for patients with HER2 positive breast tumors to predict if each patient will achieve a complete remission of the tumor so that the tumor is completely absent and cannot even be found on pathologic tissue examination.
“We aim to emerge from this short-term, high-impact effort with established cut-points, reagents, and CLIA/CAP SOPs developed and validated for our assay that would be published for general use,” said Lance Liotta, MD, PhD, study PI, and CAPMM co-founder and co-director.
During the first year of the partnership, researchers will measure HER2 activation retrospectively on previously collected cancer specimens and cross reference it with treatment outcomes. During the second year, those findings will help inform the design of prospective trials enabling real-time decision making influencing the selection of future therapy options offered to patients at Rutgers Cancer Institute and RWJBarnabas Health.
“We believe the test can have great benefit for individualizing therapies,” added, Dr. Emanuel Petricoin, PhD, Co-PI of the study and also co-founder and co-director of Mason CAPMM. “Clinical studies have supported the recent approval of two escalation strategies, unfortunately, these escalations are associated with considerable increased costs and additional risks of toxicity,” Petricoin explained.
“Therefore, careful and accurate patient risk stratification up-front, to identify which patients will benefit most from treatment, is essential to minimize risk and maximize benefit,” said Rutgers Cancer Institute’s principal investigator, Shridar Ganesan, MD, PhD, chief of molecular oncology, associate director for translational research, and Omar Boraie Chair in Genomic Science.
If validated, this predictor may help lead to development for modified or, de-escalated regimens for patients who score at low risk for recurrence or high chance of complete pathologic response Similarly, patients who score at high risk of recurrence or low chance of complete pathologic response from current standard therapy may benefit from trials exploring other treatment approaches.
“The tests will be conducted on the pretreatment biopsy tissue we have collected and directly evaluate the molecular target of the major classes of HER2 neoadjuvant therapies,” said Ganesan, a professor of medicine and pharmacology at Rutgers Robert Wood Johnson Medical School who had previously collaborated with Liotta while he was at the NIH.
“This partnership allows us to continue to provide the most advanced treatment options possible for our patients,” Ganesan added.
The team’s plan also includes a rapid deployment of the testing if it proves conclusive.
“We’ve partnered with Theralink Diagnostics, Inc. who will be responsible for the assay commercialization and expanded clinical assessment to provide an accelerated adoption and clinical use by medical oncologists,” said Emanuel Petricoin, CAPMM co-director, and a scientific advisor to the Company. “We have set up the infrastructure to quickly deploy the information and technology; if this holds true, we have a commercial partner ready to take it to the bedside as part of a patient clinical care plan and treatment decision support tool,” Petricoin explained.
“This is a smart, strategic partnership—both Rutgers and Mason each bring invaluable expertise and infrastructure to minimize time to market and maximize research impact,” said Mason College of Science dean, Fernando Miralles-Wilhelm. “Mason scientists created the protein measurement test and offer CAP/CLIA facilities that dovetail well with the existing biobank of tissue samples and preeminent therapeutic cancer treatments options offered to patients at Rutgers Cancer Institute of New Jersey,” Miralles-Wilhelm added.